Deterministic Compliance
for Healthcare AI.
Medical Device (MDR) and EU AI Act intersection is complex. Sentinel ensures your clinical algorithms are bias-free and cryptographically certified without ever touching patient PHI.
Clinical Vulnerabilities
MDR & AI Act Intersection
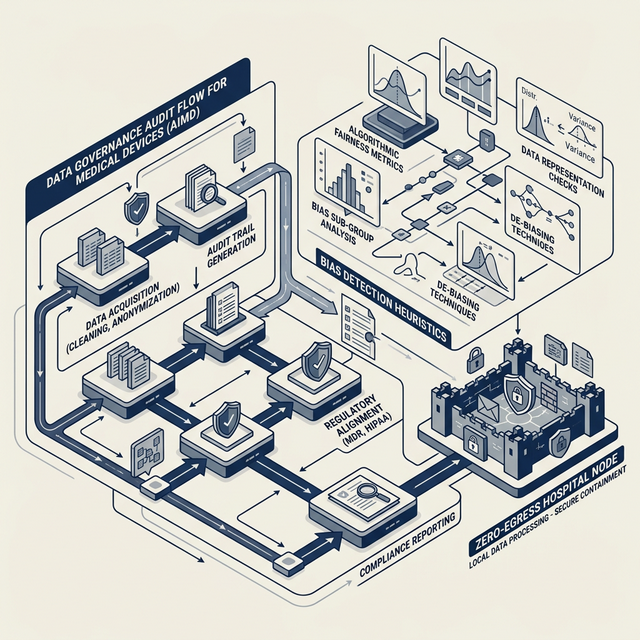
If your AI influences a clinical decision, it is classified as **High-Risk** under Annex III. Regulators demand complete transparency, yet HIPAA/GDPR forbids exposing raw patient data.
Article 10: Bias Detection Engine
Our clinical bias detection engine executes categorical distribution analysis to mitigate algorithmic health imbalance without seeing raw patient records.
Article 11: Technical File
Automated generation of Annex IV dossiers for Artificial Intelligence Medical Devices (AIMD), signed via Ed25519.
Diagnostics & Imaging
Computer vision models detecting anomalies must prove they don't suffer from demographic bias. Sentinel audits the model's structural fairness within your own clean-room.
- Automated Article 10 Bias Checks
- ISO 13485 Alignment Tracking
Clinical Decision Support
Systems recommending treatment plans require continuous monitoring. Sentinel's CI/CD Watchtower detects 'Clinical Drift' before it impacts patient outcomes.
- Real-time MDR/AI Act Drift Alerts
- Immutable D1 Ledger Certification
The Clinical Solution
Biased Algorithmic
Health Mitigation
We offer a specialized solution using Feature Distribution Heuristics to satisfy Article 10.3. It measures the entropy of categorical identifiers in your training logs while maintaining 100% data isolation.
Sovereign Health Audit.
Traditional compliance tools require you to expose model telemetry, which often inadvertently contains Protected Health Information (PHI). Sentinel operates as a compiled WebAssembly binary inside your VPC. We grade the math; we never see the patient.